|
2/29/2024 0 Comments Sodium element model
Visit CGTrader and browse more than 1 million 3D models, including 3D print and real-time assets. The internuclear distance in the gas phase is 175 pm.Īnswer: −3180 kJ/mol = −3. Model available for download in AutoCAD format. 1 Write protons, neutrons, and electrons of sodium atom. Consequently, in accordance with Equation 4.1.1, much more energy is released when 1 mol of gaseous Li +F − ion pairs is formed (−891 kJ/mol) than when 1 mol of gaseous Na +Cl − ion pairs is formed (−589 kJ/mol).Ĭalculate the amount of energy released when 1 mol of gaseous MgO ion pairs is formed from the separated ions. Here’s how you can draw the Bohr model of sodium step by step. It cannot be maintained in an inert atmosphere and contact with water and other substances with which sodium reacts should be avoided.\( \newcommand/mol\right )=-891\ kJ/mol \) īecause Li + and F − are smaller than Na + and Cl − (see Figure 3.2.7 ), the internuclear distance in LiF is shorter than in NaCl. Sodium metal should be handled with great care. Thirteen isotopes of sodium are recognized. The importance of common salt to animal nutrition has been recognized since prehistoric times.Īmong the many compounds that are of the greatest industrial importance are common salt (NaCl), soda ash (Na 2CO 3), baking soda (NaHCO 3), caustic soda (NaOH), Chile saltpeter (NaNO 3), di- and tri-sodium phosphates, sodium thiosulfate (hypo, Na 2S 2O 3 Soap is generally a sodium salt of certain fatty acids. Sodium compounds are important to the paper, glass, soap, textile, petroleum, chemical, and metal industries. The metal may be used to improve the structure of certain alloys, descale metal, and purify molten metals.Īn alloy of sodium with potassium, NaK, is an important heat transfer agent. Metallic sodium is vital in the manufacture of esters and in the preparation of organic compounds. It normally does not ignite in air at temperatures below 115☌. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. It may or may not ignite spontaneously on water, depending on the amount of oxide and metal exposed to the water. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Decomposition in water results in the evolution of hydrogen and the formation of the hydroxide. Sodium is a soft, bright, silvery metal which floats on water. Sodium was discovered and first isolated by Sir Humphrey Davy in 1807. Sodium, like every reactive element, is never found free in nature. The sodium atom has a radius of 185.8 pm and a Van der Waals radius of 227 pm. It is highly reactive, burns with a yellow flame, oxidizes in air and. It is the most abundant of the alkali metals. Sodium is a soft, waxy, silvery reactive metal belonging to the alkali metals that is abundant in natural compounds (especially halite). It is the sixth most abundant element on earth with 2.6 of the earths crust. 
The most common compound is sodium chloride (table salt), but it occurs in many other minerals, such as soda niter, cryolite, amphibole, zeolite, etc. Sodium is a chemical element in the periodic table that has the symbol Na ( Natrium in Latin) and atom number 11. This method is much cheaper than that of electrolyzing sodium hydroxide, as was used several years ago. It is now obtained commercially by the electrolysis of absolutely dry fused sodium chloride. Sodium is the fourth most abundant element on earth, comprising about 2.6% of the earth's crust it is the most abundant of the alkali group of metals. It has a complete octet of outer-shell electrons and is a noble gas. Sodium is a soft, waxy, silvery reactive. Element number 18, argon, Ar, atomic mass 39.95, brings us to the end of the third period of the abbreviated periodic table. The D lines of sodium are among the most prominent in the solar spectrum. Sodium is a chemical element in the periodic table that has the symbol Na (Natrium in Latin) and atom number 11. Sodium is present in fair abundance in the sun and stars. Sodium is a chemical element of the periodic table with chemical symbol Na and atomic number 11 with an atomic weight of 22.9898 u and is classed as alkali. Long recognized in compounds, sodium was first isolated by Davy in 1807 by electrolysis of caustic soda. 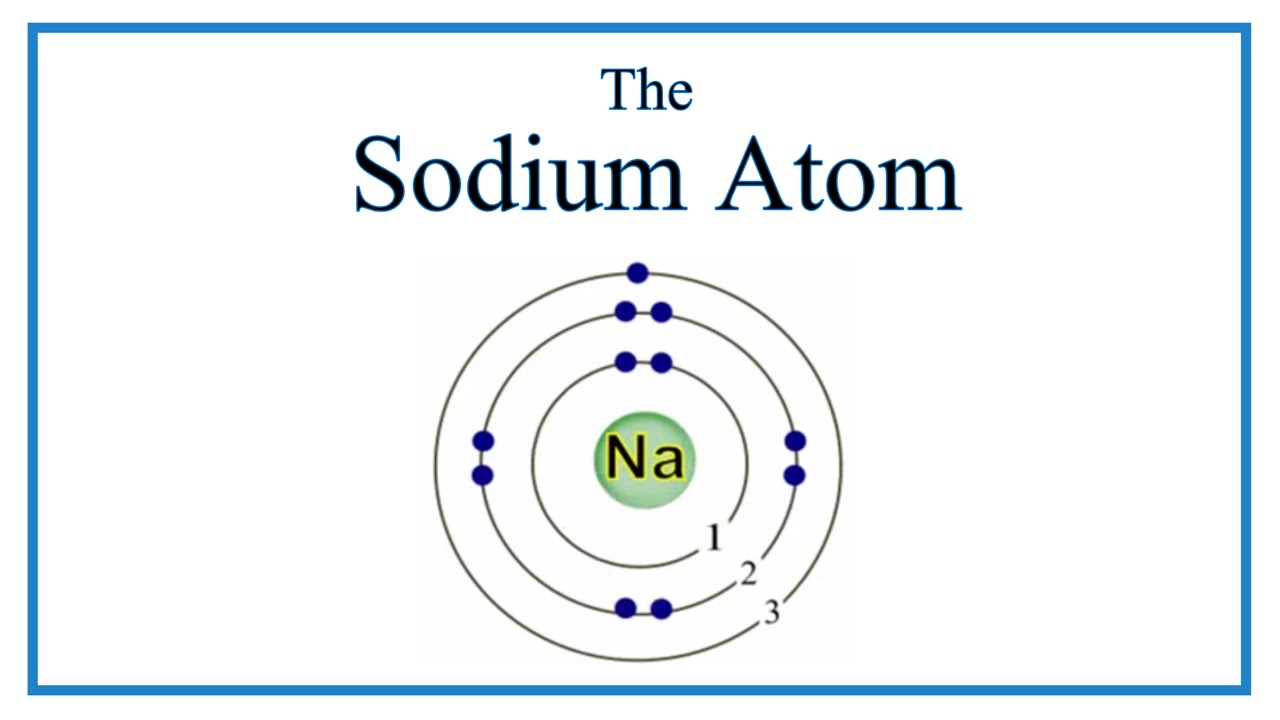
Sodium is a soft, bright, silvery metal which floats on water.įrom the English word, soda Medieval Latin, sodanum: a headache remedy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
